BPV are UK validation experts offering autoclave validation, thermal mapping, and equipment validation services, as well as validation equipment hire and validation consulting, commissioning, qualification and validation (CQV) services. We use our years of experience to support regulated industries in staying compliant and reliable, with processes and equipment that consistently meet regulatory and quality requirements.
By adhering to the core principles of ISO 9001, BPV establishes a strong
foundation for effective quality management, enhancing customer satisfaction, and fostering a culture of excellence that drives long-term success in providing top-tier validation services.
As your EU and UK validation provider and partner, we are committed to helping your company achieve its goals while maintaining regulatory compliance. We offer end-to-end support, from project initiation to validation planning, and optimisation.
As part of our commitment to streamline our client's validation processes, we've developed our own cGMP-compliant validation protocols. These proven protocols can be adapted to your format if needed, or you can use your own. We're happy to work with either.
BPV is your UK and EU validation service provider, offering:
Sterilisation Validation
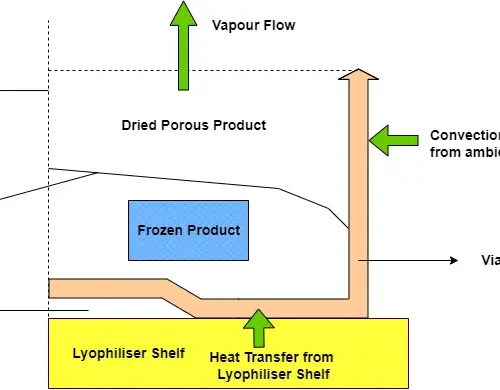
BPV specialises in sterilisation validation services, guaranteeing the safe, reliable, and compliant operation of sterilisation processes in line with regulatory requirements.
Thermal Mapping
BPV ensures GMP compliance in maintaining thermal uniformity and safeguarding product quality in pharmaceutical warehouses, cold storage facilities, and controlled temperature units (CTUs).
Steam Quality Testing
BPV specialises in complete steam quality testing services for life science industries. This ensures regulatory compliance, early issue detection, and optimal equipment performance through expert evaluation and industry-standard testing methods.
Validation Equipment Hire
BPV offers validation equipment hire services, including calibrated Kaye Validators and accessories. This provides a flexible and cost-effective solution for pharmaceutical manufacturing validation projects, promoting operational efficiency.
Validation Consulting
BPV provides expert validation consulting tailored to the unique needs of life science companies. Our team offers unparalleled guidance throughout your validation journey, ensuring regulatory compliance and patient safety.
CSV validation services
BPV supplies thorough Computer Systems Validation (CSV) services, encompassing all validation stages, to ensure strict compliance with GxP standards and GAMP 5 methodologies.
Process Validation
We offer comprehensive process validation services, including development, execution, and periodic verification to ensure safe and consistent production of high-quality pharmaceutical products throughout their lifecycle.
Cleaning Validation
BPV executes cleaning validation studies for pharmaceutical manufacturers, including manual cleaning processes and CIP validation. This ensures regulatory compliance and promotes patient safety.
Contact Us
We’re here to help with your validation needs. Whether you have questions about our validation services, need expert advice, or want to discuss a project, our team is ready to assist you.
We look forward to hearing from you and helping you achieve your validation goals.